 “Current pharmacotherapy of Parkinson’s disease (PD) is symptomatic and palliative, with levodopa/carbidopa therapy remaining the prime treatment, and nevertheless, being unable to modulate the progression of the neurodegeneration. No available treatment for PD can enhance the patient’s life-quality by regressing this diseased state.
“Current pharmacotherapy of Parkinson’s disease (PD) is symptomatic and palliative, with levodopa/carbidopa therapy remaining the prime treatment, and nevertheless, being unable to modulate the progression of the neurodegeneration. No available treatment for PD can enhance the patient’s life-quality by regressing this diseased state.
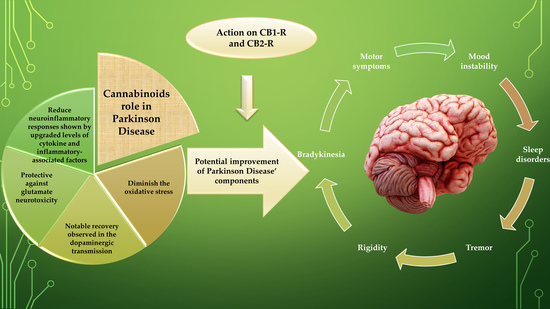
Various studies have encouraged the enrichment of treatment possibilities by discovering the association of the effects of the endocannabinoid system (ECS) in PD.
These reviews delineate the reported evidence from the literature on the neuromodulatory role of the endocannabinoid system and expression of cannabinoid receptors in symptomatology, cause, and treatment of PD progression, wherein cannabinoid (CB) signalling experiences alterations of biphasic pattern during PD progression.
Endocannabinoids regulate the basal ganglia neuronal circuit pathways, synaptic plasticity, and motor functions via communication with dopaminergic, glutamatergic, and GABAergic signalling systems bidirectionally in PD.
Further, gripping preclinical and clinical studies demonstrate the context regarding the cannabinoid compounds, which is supported by various evidence (neuroprotection, suppression of excitotoxicity, oxidative stress, glial activation, and additional benefits) provided by cannabinoid-like compounds (much research addresses the direct regulation of cannabinoids with dopamine transmission and other signalling pathways in PD).
More data related to endocannabinoids efficacy, safety, and pharmacokinetic profiles need to be explored, providing better insights into their potential to ameliorate or even regress PD.”
https://pubmed.ncbi.nlm.nih.gov/32872273/
https://www.mdpi.com/1422-0067/21/17/6235
 “Prostate cancer is a major cause of death among men worldwide.
“Prostate cancer is a major cause of death among men worldwide.


 “Previous preclinical studies have demonstrated that cannabidiol (CBD) and cannabigerol (CBG), two non-psychotomimetic phytocannabinoids from Cannabis sativa, induce neuroprotective effects on toxic and neurodegenerative processes.
“Previous preclinical studies have demonstrated that cannabidiol (CBD) and cannabigerol (CBG), two non-psychotomimetic phytocannabinoids from Cannabis sativa, induce neuroprotective effects on toxic and neurodegenerative processes. “Cannabidiol (CBD) is considered a non-psychoactive, antioxidant, and anti-inflammatory compound derived from the Cannabis sativa plant.
“Cannabidiol (CBD) is considered a non-psychoactive, antioxidant, and anti-inflammatory compound derived from the Cannabis sativa plant. “Cannabidiol (CBD) is a non-intoxicating phytocannabinoid from cannabis sativa that has demonstrated anti-inflammatory effects in several inflammatory conditions including arthritis.
“Cannabidiol (CBD) is a non-intoxicating phytocannabinoid from cannabis sativa that has demonstrated anti-inflammatory effects in several inflammatory conditions including arthritis. “Staphylococcal enterotoxin B (SEB) is a potent activator of Vβ8+T-cells resulting in the clonal expansion of ∼30% of the T-cell pool. Consequently, this leads to the release of inflammatory cytokines, toxic shock, and eventually death.
“Staphylococcal enterotoxin B (SEB) is a potent activator of Vβ8+T-cells resulting in the clonal expansion of ∼30% of the T-cell pool. Consequently, this leads to the release of inflammatory cytokines, toxic shock, and eventually death. “Staphylococcal enterotoxin‐B (SEB) is one of the most potent bacterial superantigens that exerts profound toxic effects by inducing cytokine storm. When SEB is inhaled, it can cause Acute Respiratory Distress Syndrome (ARDS), which is often fatal and currently there are no effective treatment modalities.
“Staphylococcal enterotoxin‐B (SEB) is one of the most potent bacterial superantigens that exerts profound toxic effects by inducing cytokine storm. When SEB is inhaled, it can cause Acute Respiratory Distress Syndrome (ARDS), which is often fatal and currently there are no effective treatment modalities. “Coronavirus disease-2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is evolving across the world and new treatments are urgently needed as with vaccines to prevent the illness and stem the contagion. The virus affects not only the lungs but also other tissues, thus lending support to the idea that COVID-19 is a systemic disease. The current vaccine and treatment development strategies ought to consider such systems medicine perspectives rather than a narrower focus on the lung infection only.
“Coronavirus disease-2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is evolving across the world and new treatments are urgently needed as with vaccines to prevent the illness and stem the contagion. The virus affects not only the lungs but also other tissues, thus lending support to the idea that COVID-19 is a systemic disease. The current vaccine and treatment development strategies ought to consider such systems medicine perspectives rather than a narrower focus on the lung infection only.