“Modulation of the endogenous cannabinoid system has been suggested as a potential anticancer strategy.
In the search for novel and less toxic therapeutic options, structural modifications of the endocannabinoid anandamide and the synthetic derivative of oleic acid, Minerval (HU-600), were done to obtain 2-hydroxy oleic acid ethanolamide (HU-585), which is an HU-600 derivative with the anandamide side chain.
We showed that treatment of SK-N-SH neuroblastoma cells with HU-585 induced a better anti-tumorigenic effect in comparison to HU-600 as evidenced by 3-[4,5-dimethylthiazole-2-yl]-2,5-diphenyltetrazolium bromide assay, colony-forming assay, and migration assay. Moreover, HU-585 demonstrated pro-apoptotic properties shown by increased levels of activated caspase-3 following treatment and a better senescence induction effect in comparison to HU-600, as demonstrated by increased activity of lysosomal β-galactosidase. Finally, we observed that combined treatment of HU-585 with the senolytic drugs ABT-263 in vitro, and ABT-737 in vivo resulted in enhanced anti-proliferative effects and reduced neuroblastoma xenograft growth in comparison to treatment with HU-585 alone.
Based on these results, we suggest that HU-585 is a pro-apoptotic and senescence-inducing compound, better than HU-600. Hence, it may be a beneficial option for the treatment of resistant neuroblastoma especially when combined with senolytic drugs that enhance its anti-tumorigenic effects.”
https://pubmed.ncbi.nlm.nih.gov/35884854/
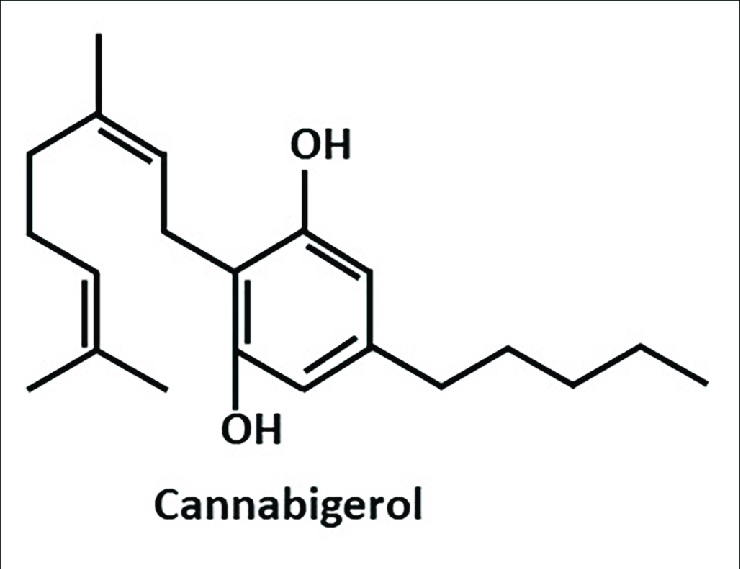
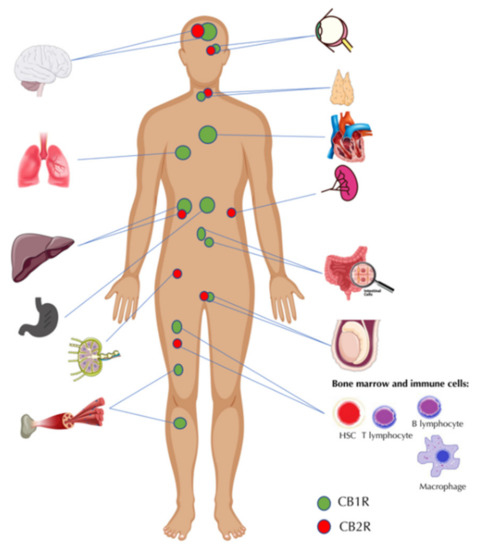
“The cannabinoids are a group of more than 100 chemically related compounds found in the marijuana plant Cannabis sativa, that have been found to possess diverse pharmacological activities in cancer, including cytostatic, apoptotic, and antiangiogenic effects. Tetrahydrocannabinol (THC), the main psychoactive constituent in Cannabis sativa, acts mainly through the activation of specific cannabinoid receptors CB1 and CB2 and thus mimics the binding of the animal endogenous cannabinoids (named endocannabinoids).”