 “Acute Respiratory Distress Syndrome (ARDS) causes up to 40% mortality in humans and is difficult to treat. ARDS is also one of the major triggers of mortality associated with coronavirus-induced disease (COVID-19). We used a mouse model of ARDS induced by Staphylococcal enterotoxin B (SEB), which triggers 100% mortality, to investigate the mechanisms through which Δ9-tetrahydrocannabinol (THC) attenuates ARDS.
“Acute Respiratory Distress Syndrome (ARDS) causes up to 40% mortality in humans and is difficult to treat. ARDS is also one of the major triggers of mortality associated with coronavirus-induced disease (COVID-19). We used a mouse model of ARDS induced by Staphylococcal enterotoxin B (SEB), which triggers 100% mortality, to investigate the mechanisms through which Δ9-tetrahydrocannabinol (THC) attenuates ARDS.
SEB was used to trigger ARDS in C3H mice. These mice were treated with THC and analyzed for survival, ARDS, cytokine storm, and metabolome. Additionally, cells isolated from the lungs were used to perform single-cell RNA sequencing and transcriptome analysis. A database analysis of human COVID-19 patients was also performed to compare the signaling pathways with SEB-mediated ARDS.
The treatment of SEB-mediated ARDS mice with THC led to a 100% survival, decreased lung inflammation, and the suppression of cytokine storm. This was associated with immune cell apoptosis involving the mitochondrial pathway, as suggested by single-cell RNA sequencing. A transcriptomic analysis of immune cells from the lungs revealed an increase in mitochondrial respiratory chain enzymes following THC treatment. In addition, metabolomic analysis revealed elevated serum concentrations of amino acids, lysine, n-acetyl methionine, carnitine, and propionyl L-carnitine in THC-treated mice. THC caused the downregulation of miR-185, which correlated with an increase in the pro-apoptotic gene targets. Interestingly, the gene expression datasets from the bronchoalveolar lavage fluid (BALF) of human COVID-19 patients showed some similarities between cytokine and apoptotic genes with SEB-induced ARDS.
Collectively, this study suggests that the activation of cannabinoid receptors may serve as a therapeutic modality to treat ARDS associated with COVID-19.”

 “Coronavirus disease-2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is evolving across the world and new treatments are urgently needed as with vaccines to prevent the illness and stem the contagion. The virus affects not only the lungs but also other tissues, thus lending support to the idea that COVID-19 is a systemic disease. The current vaccine and treatment development strategies ought to consider such systems medicine perspectives rather than a narrower focus on the lung infection only.
“Coronavirus disease-2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is evolving across the world and new treatments are urgently needed as with vaccines to prevent the illness and stem the contagion. The virus affects not only the lungs but also other tissues, thus lending support to the idea that COVID-19 is a systemic disease. The current vaccine and treatment development strategies ought to consider such systems medicine perspectives rather than a narrower focus on the lung infection only. “Coronavirus disease 2019 (COVID-19) is spreading fast all around the world with more than fourteen millions of detected infected cases and more than 600.000 deaths by 20th July 2020. While scientist are working to find a vaccine, current epidemiological data shows that the most common comorbidities for patients with the worst prognosis, hypertension and diabetes, are often treated with angiotensin converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs).
“Coronavirus disease 2019 (COVID-19) is spreading fast all around the world with more than fourteen millions of detected infected cases and more than 600.000 deaths by 20th July 2020. While scientist are working to find a vaccine, current epidemiological data shows that the most common comorbidities for patients with the worst prognosis, hypertension and diabetes, are often treated with angiotensin converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs). “So far, no vaccine has been successfully developed and there is no effective treatment of COVID-19.
“So far, no vaccine has been successfully developed and there is no effective treatment of COVID-19. “Acute Respiratory Distress Syndrome (ARDS) is a life-threatening complication that can ensue following Staphylococcus aureus infection. The enterotoxin produced by these bacteria (SEB) acts as a superantigen thereby activating a large proportion of T cells leading to cytokine storm and severe lung injury.
“Acute Respiratory Distress Syndrome (ARDS) is a life-threatening complication that can ensue following Staphylococcus aureus infection. The enterotoxin produced by these bacteria (SEB) acts as a superantigen thereby activating a large proportion of T cells leading to cytokine storm and severe lung injury. “Identifying candidate drugs effective in the new coronavirus disease 2019 (Covid-19) is crucial,
“Identifying candidate drugs effective in the new coronavirus disease 2019 (Covid-19) is crucial, 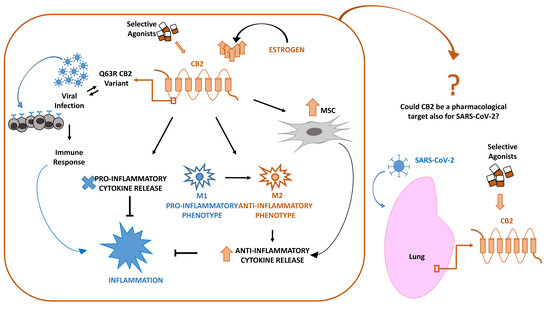

 “Coronavirus disease-2019 (COVID-19), caused by Severe Acute Respiratory Syndrome coronoavirus-2 (SARS-CoV2) has emerged as a global pandemic, which was first reported in Wuhan, China. Recent reports have suggested that acute infection is associated with a cytokine superstorm, which contributes to the symptoms of fever, cough, muscle pain and in severe cases bilateral interstitial pneumonia characterized by ground glass opacity and focal chest infiltrates that can be visualized on computerized tomography scans. Currently, there are no effective antiviral drugs or vaccines against SARS-CoV2. In the recent issue of BBI, Zhang et al. thoroughly summarized the current status of potential therapeutic strategies for COVID-19. One of them, anti-IL6 receptor (Tocilizumab) antibody, resulted in clearance of lung consolidation and recovery in 90% of the 21 treated patients. Although promising, it has also produced adverse effects like pancreatitis and hypertriglyceridemia, which make it imperative to explore effective alternative anti-inflammatory strategies. Here, we intend to highlight the potential effects of cannabinoids, in particular, the non-psychotropic cannabidiol (CBD), that has shown beneficial anti-inflammatory effects in pre-clinical models of various chronic inflammatory diseases and is FDA approved for seizure reduction in children with intractable epilepsy.
“Coronavirus disease-2019 (COVID-19), caused by Severe Acute Respiratory Syndrome coronoavirus-2 (SARS-CoV2) has emerged as a global pandemic, which was first reported in Wuhan, China. Recent reports have suggested that acute infection is associated with a cytokine superstorm, which contributes to the symptoms of fever, cough, muscle pain and in severe cases bilateral interstitial pneumonia characterized by ground glass opacity and focal chest infiltrates that can be visualized on computerized tomography scans. Currently, there are no effective antiviral drugs or vaccines against SARS-CoV2. In the recent issue of BBI, Zhang et al. thoroughly summarized the current status of potential therapeutic strategies for COVID-19. One of them, anti-IL6 receptor (Tocilizumab) antibody, resulted in clearance of lung consolidation and recovery in 90% of the 21 treated patients. Although promising, it has also produced adverse effects like pancreatitis and hypertriglyceridemia, which make it imperative to explore effective alternative anti-inflammatory strategies. Here, we intend to highlight the potential effects of cannabinoids, in particular, the non-psychotropic cannabidiol (CBD), that has shown beneficial anti-inflammatory effects in pre-clinical models of various chronic inflammatory diseases and is FDA approved for seizure reduction in children with intractable epilepsy.
 “Cannabis Indica Speeds up Recovery from Coronavirus Severe acute respiratory syndrome (SARS) is a viral respiratory disease caused by the SARS coronavirus (SARS-CoV).
“Cannabis Indica Speeds up Recovery from Coronavirus Severe acute respiratory syndrome (SARS) is a viral respiratory disease caused by the SARS coronavirus (SARS-CoV).