
“Phytocannabinoids possess a wide range of immune regulatory properties, mediated by the endocannabinoid system.
Monocyte/macrophage innate immune cells express endocannabinoid receptors. Dysregulation of macrophage function is involved in the pathogenesis of different inflammatory diseases, including inflammatory bowel disease.
In our research, we aimed to evaluate the effects of the phytocannabinoids D9 tetrahydrocannabinol (THC) and cannabidiol (CBD) on macrophage activation.
Macrophages from young and aged C57BL/6 mice were activated in vitro in the presence of pure cannabinoids or cannabis extracts. The phenotype of the cells, nitric oxide (NO•) secretion, and cytokine secretion were examined. In addition, these treatments were administered to murine colitis model. The clinical statuses of mice, levels of colon infiltrating macrophages, and inflammatory cytokines in the blood, were evaluated.
We demonstrated inhibition of macrophage NO• and cytokine secretion and significant effects on expression of cell surface molecules. In the murine model, clinical scores were improved and macrophage colon infiltration reduced following treatment. We identified higher activity of cannabis extracts as compared with pure cannabinoids. Each treatment had a unique effect on cytokine composition.
Overall, our results establish that the effects of cannabinoid treatments differ. A better understanding of the reciprocal relationship between cannabinoids and immunity is essential to design targeted treatment strategies.”
https://pubmed.ncbi.nlm.nih.gov/35892693/
“Overall, our results indicate both similarities and differences between the impact of CBD- and THC-based drugs. Although all the tested treatments had an anti-inflammatory effect, their specific effects (for example, on phenotype of the cells and on cytokine production) differed. These differences may influence the clinical outcome of the treatment. We were surprised to find very similar anti-inflammatory results for the two cannabis extracts, which had diverse content of THC and CBD. This could suggest that THC/CBD content may not be the best indicator for anti-inflammatory properties of a cannabis-based drug. These results highlight the need to expand the research on the interplay between cannabinoids and other phytochemicals in the cannabis extracts. A better understanding of the effects of each molecule and the synergism between these molecules on the immune response will assist physicians to provide the best possible individually targeted treatment for their patients and will allow the design of new treatments.”



 “Inflammatory bowel diseases (IBDs) are chronic, idiopathic, inflammatory, gastrointestinal disorders.
“Inflammatory bowel diseases (IBDs) are chronic, idiopathic, inflammatory, gastrointestinal disorders. 
 “The Endocannabinoid System (ECS) is primarily responsible for maintaining homeostasis, a balance in internal environment (temperature, mood, and immune system) and energy input and output in living, biological systems.
“The Endocannabinoid System (ECS) is primarily responsible for maintaining homeostasis, a balance in internal environment (temperature, mood, and immune system) and energy input and output in living, biological systems. 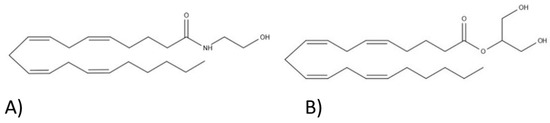
 “The relevance and incidence of intestinal bowel diseases (IBD) have been increasing over the last 50 years and the current therapies are characterized by severe side effects, making essential the development of new strategies that combine efficacy and safety in the management of human IBD. Herbal products are highly considered in research aimed at discovering new approaches for IBD therapy and, among others,
“The relevance and incidence of intestinal bowel diseases (IBD) have been increasing over the last 50 years and the current therapies are characterized by severe side effects, making essential the development of new strategies that combine efficacy and safety in the management of human IBD. Herbal products are highly considered in research aimed at discovering new approaches for IBD therapy and, among others,  “Medical cannabis and individual cannabinoids, such as tetrahydrocannabinol (THC) and cannabidiol (CBD), are receiving growing attention in both the media and the scientific literature. The Cannabis plant, however, produces over 100 different cannabinoids, and cannabigerol (CBG) serves as the precursor molecule for the most abundant phytocannabinoids.
“Medical cannabis and individual cannabinoids, such as tetrahydrocannabinol (THC) and cannabidiol (CBD), are receiving growing attention in both the media and the scientific literature. The Cannabis plant, however, produces over 100 different cannabinoids, and cannabigerol (CBG) serves as the precursor molecule for the most abundant phytocannabinoids.
