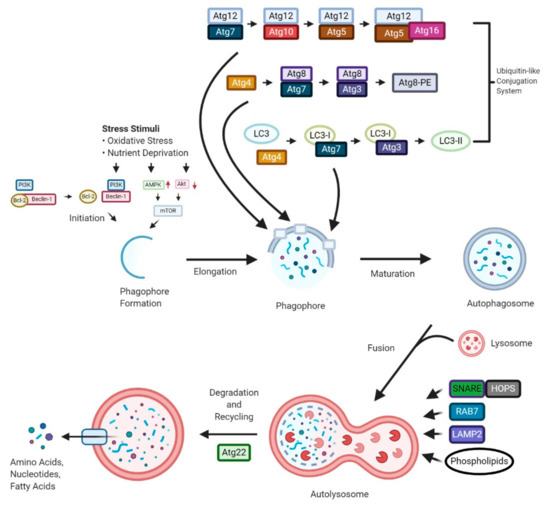
 “Autophagy is a “self-degradation” process whereby malfunctioned cytoplasmic constituents and protein aggregates are engulfed by a vesicle called the autophagosome, and subsequently degraded by the lysosome. Autophagy plays a crucial role in sustaining protein homeostasis and can be an alternative source of energy under detrimental circumstances. Studies have demonstrated a paradoxical function for autophagy in cancer, displaying both tumour suppressive and tumour promotive roles. In early phases of tumour development autophagy promotes cancer cell death. In later phases, autophagy enables cancer cells to survive and withstand therapy.
“Autophagy is a “self-degradation” process whereby malfunctioned cytoplasmic constituents and protein aggregates are engulfed by a vesicle called the autophagosome, and subsequently degraded by the lysosome. Autophagy plays a crucial role in sustaining protein homeostasis and can be an alternative source of energy under detrimental circumstances. Studies have demonstrated a paradoxical function for autophagy in cancer, displaying both tumour suppressive and tumour promotive roles. In early phases of tumour development autophagy promotes cancer cell death. In later phases, autophagy enables cancer cells to survive and withstand therapy.
Cannabinoids, which are derivatives of the Cannabis sativa L. plant, have shown to be associated with autophagy induction in cells. There is an emerging interest in studying the signalling pathways involved in cannabinoid-induced autophagy and their potential application in anticancer therapies. In this review, the molecular mechanisms involved in the autophagy degradation process will be discussed. This review also highlights a role for autophagy in cancer progression, with cannabinoid-induced autophagy presenting a novel strategy for anticancer therapy.”
https://pubmed.ncbi.nlm.nih.gov/33802014/


 “Purpose: Hodgkin lymphoma (HL) is the fourth most frequent cancer diagnosis among pregnant females. A multidisciplinary team is mandatory to obtain the best treatment and prognosis for the mother and for the baby. Here, we present the case of a patient diagnosed with HL and its evolution during 2 pregnancies.
“Purpose: Hodgkin lymphoma (HL) is the fourth most frequent cancer diagnosis among pregnant females. A multidisciplinary team is mandatory to obtain the best treatment and prognosis for the mother and for the baby. Here, we present the case of a patient diagnosed with HL and its evolution during 2 pregnancies. “Breast cancer is the leading cause of cancer-related death in women worldwide. In the last years, cannabinoids have gained attention in the clinical setting and clinical trials with cannabinoid-based preparations are underway. However, contradictory anti-tumour properties have also been reported. Thus, the elucidation of the molecular mechanisms behind their anti-tumour efficacy is crucial to better understand its therapeutic potential.
“Breast cancer is the leading cause of cancer-related death in women worldwide. In the last years, cannabinoids have gained attention in the clinical setting and clinical trials with cannabinoid-based preparations are underway. However, contradictory anti-tumour properties have also been reported. Thus, the elucidation of the molecular mechanisms behind their anti-tumour efficacy is crucial to better understand its therapeutic potential. “Cannabis was extensively utilized for its medicinal properties till the 19th century. A steep decline in its medicinal usage was observed later due to its emergence as an illegal recreational drug. Advances in technology and scientific findings led to the discovery of delta-9-tetrahydrocannabinol (THC), the primary psychoactive compound of cannabis, that further led to the discovery of endogenous cannabinoids system consisting of G-protein-coupled receptors – cannabinoid receptor 1 and cannabinoid receptor 2 along with their ligands, mainly anandamide and 2-arachidonoylglycerol. Endocannabinoid (EC) is shown to be a modulator not only for physiological functions but also for the immune system, endocrine network, and central nervous system. Medicinal research and meta-data analysis over the last few decades have shown a significant potential for both THC and cannabidiol (CBD) to exert palliative effects. People suffering from many forms of advanced stages of cancers undergo chemotherapy-induced nausea and vomiting followed by severe and chronic neuropathic pain and weight loss. THC and CBD exhibit effective analgesic, anxiolytic, and appetite-stimulating effect on patients suffering from cancer. Drugs currently available in the market to treat such chemotherapy-induced cancer-related ailments are Sativex (GW Pharmaceutical), Dronabinol (Unimed Pharmaceuticals), and Nabilone (Valeant Pharmaceuticals). Apart from exerting palliative effects, THC also shows promising role in the treatment of cancer growth, neurodegenerative diseases (multiple sclerosis and Alzheimer’s disease), and alcohol addiction and hence should be exploited for potential benefits. The current review discusses the nature and role of CB receptors, specific applications of cannabinoids, and major studies that have assessed the role of cannabinoids in cancer management.”
“Cannabis was extensively utilized for its medicinal properties till the 19th century. A steep decline in its medicinal usage was observed later due to its emergence as an illegal recreational drug. Advances in technology and scientific findings led to the discovery of delta-9-tetrahydrocannabinol (THC), the primary psychoactive compound of cannabis, that further led to the discovery of endogenous cannabinoids system consisting of G-protein-coupled receptors – cannabinoid receptor 1 and cannabinoid receptor 2 along with their ligands, mainly anandamide and 2-arachidonoylglycerol. Endocannabinoid (EC) is shown to be a modulator not only for physiological functions but also for the immune system, endocrine network, and central nervous system. Medicinal research and meta-data analysis over the last few decades have shown a significant potential for both THC and cannabidiol (CBD) to exert palliative effects. People suffering from many forms of advanced stages of cancers undergo chemotherapy-induced nausea and vomiting followed by severe and chronic neuropathic pain and weight loss. THC and CBD exhibit effective analgesic, anxiolytic, and appetite-stimulating effect on patients suffering from cancer. Drugs currently available in the market to treat such chemotherapy-induced cancer-related ailments are Sativex (GW Pharmaceutical), Dronabinol (Unimed Pharmaceuticals), and Nabilone (Valeant Pharmaceuticals). Apart from exerting palliative effects, THC also shows promising role in the treatment of cancer growth, neurodegenerative diseases (multiple sclerosis and Alzheimer’s disease), and alcohol addiction and hence should be exploited for potential benefits. The current review discusses the nature and role of CB receptors, specific applications of cannabinoids, and major studies that have assessed the role of cannabinoids in cancer management.” “Cannabidiol (CBD) has been shown to slow cancer cell growth and is toxic to human glioblastoma cell lines. Thus, CBD could be an effective therapeutic for glioblastoma.
“Cannabidiol (CBD) has been shown to slow cancer cell growth and is toxic to human glioblastoma cell lines. Thus, CBD could be an effective therapeutic for glioblastoma.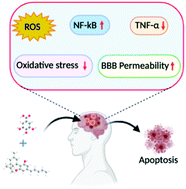
 “Considering the advantages of using medicinal herbs as supplementary treatments to sensitize conventional anti-cancer drugs, studying functional mechanisms and regulatory effects of Echinacea purpurea (as a non-cannabinoid plant)
“Considering the advantages of using medicinal herbs as supplementary treatments to sensitize conventional anti-cancer drugs, studying functional mechanisms and regulatory effects of Echinacea purpurea (as a non-cannabinoid plant) 


 “Cannabis sativa contains more than 500 constituents, yet the anticancer properties of the vast majority of cannabis compounds remains unknown. We aimed to identify cannabis compounds and their combinations presenting cytotoxicity against bladder urothelial carcinoma (UC), the most common urinary system cancer.
“Cannabis sativa contains more than 500 constituents, yet the anticancer properties of the vast majority of cannabis compounds remains unknown. We aimed to identify cannabis compounds and their combinations presenting cytotoxicity against bladder urothelial carcinoma (UC), the most common urinary system cancer. “Derivatives of the plant Cannabis sativa have been used for centuries for both medical and recreational purposes, as well as industrial. The first proof of its medicinal use comes from ancient China, although there is evidence of its earlier utilization in Europe and Asia. In the 19th century, European practitioners started to employ cannabis extracts to treat tetanus, convulsions, and mental diseases and, in 1851, cannabis made its appearance in the Pharmacopoeia of the United States as an analgesic, hypnotic and anticonvulsant. It was only in 1937 that the Marijuana Tax Act prohibited the use of this drug in the USA. The general term Cannabis is commonly used by the scientific and scholar community to indicate derivatives of the plant Cannabis sativa. The word cannabinoid is a term describing chemical compounds that are either derivate of Cannabis (phytocannabinoids) or artificial analogues (synthetic) or are produced endogenously by the body (endocannabinoids). A more casual term “marijuana” or “weed”, a compound derived from dried Cannabis flower tops and leaves, has progressively superseded the term cannabis when referred to its recreational use. The 2018 World health organisation (WHO) data suggest that nearly 2.5% of the global population (147 million) uses marijuana and some countries, such as Canada and Uruguay, have already legalised it. Due to its controversial history, the medicinal use of cannabinoids has always been a centre of debate. The isolation and characterisation of Δ9 tetrahydrocannabinol (THC), the major psychoactive component of cannabis and the detection of two human cannabinoid receptor (CBRs) molecules renewed interest in the medical use of cannabinoids, boosting research and commercial heed in this sector. Some cannabinoid-based drugs have been approved as medications, mainly as antiemetic, antianorexic, anti-seizure remedies and in cancer and multiple sclerosis patients’ palliative care. Nevertheless, due to the stigma commonly associated with these compounds, cannabinoids’ potential in the treatment of conditions such as cancer is still largely unknown and therefore underestimated.”
“Derivatives of the plant Cannabis sativa have been used for centuries for both medical and recreational purposes, as well as industrial. The first proof of its medicinal use comes from ancient China, although there is evidence of its earlier utilization in Europe and Asia. In the 19th century, European practitioners started to employ cannabis extracts to treat tetanus, convulsions, and mental diseases and, in 1851, cannabis made its appearance in the Pharmacopoeia of the United States as an analgesic, hypnotic and anticonvulsant. It was only in 1937 that the Marijuana Tax Act prohibited the use of this drug in the USA. The general term Cannabis is commonly used by the scientific and scholar community to indicate derivatives of the plant Cannabis sativa. The word cannabinoid is a term describing chemical compounds that are either derivate of Cannabis (phytocannabinoids) or artificial analogues (synthetic) or are produced endogenously by the body (endocannabinoids). A more casual term “marijuana” or “weed”, a compound derived from dried Cannabis flower tops and leaves, has progressively superseded the term cannabis when referred to its recreational use. The 2018 World health organisation (WHO) data suggest that nearly 2.5% of the global population (147 million) uses marijuana and some countries, such as Canada and Uruguay, have already legalised it. Due to its controversial history, the medicinal use of cannabinoids has always been a centre of debate. The isolation and characterisation of Δ9 tetrahydrocannabinol (THC), the major psychoactive component of cannabis and the detection of two human cannabinoid receptor (CBRs) molecules renewed interest in the medical use of cannabinoids, boosting research and commercial heed in this sector. Some cannabinoid-based drugs have been approved as medications, mainly as antiemetic, antianorexic, anti-seizure remedies and in cancer and multiple sclerosis patients’ palliative care. Nevertheless, due to the stigma commonly associated with these compounds, cannabinoids’ potential in the treatment of conditions such as cancer is still largely unknown and therefore underestimated.”