
Case presentation: Herein we present the case of a 21-year-old female Caucasian patient, with free history, diagnosed with HL stage IIB. The patient started first line chemotherapy and radiotherapy, with incomplete remission. She refused any other treatment. Five years later, the patient became pregnant and was offered chemotherapy in the 2nd trimester of pregnancy, that she refused, and delivered by C-section at 37 weeks. In the same year, the patient became pregnant again and was proposed termination of pregnancy, that she also refused. The MRI scan revealed progression of HL and she was admitted in the hospital several times for altered general condition, respiratory infections and increased need of painkillers including opioids.
At 26 weeks of pregnancy, the patient began on her own a treatment with pure cannabis. Her pain and general status got better and the tumor tissue decreased.
She delivered by C-section at 34 weeks a boy that presented in the first 24 h postpartum a withdrawal syndrome and intestinal invagination, requiring care in NICU and surgery with bowel resection.
Conclusion: Therefore, we can conclude that cannabis could be part of oncological treatment. No other case like this, as far as we know, has been previously reported.”

 “The cannabis plant (Cannabis sativa L.) produces an estimated 545 chemical compounds of different biogenetic classes. In addition to economic value, many of these phytochemicals have medicinal and physiological activity. The plant is most popularly known for its two most-prominent and most-studied secondary metabolites-Δ9-tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD). Both Δ9-THC and CBD have a wide therapeutic window across many ailments and form part of a class of secondary metabolites called cannabinoids-of which approximately over 104 exist.
“The cannabis plant (Cannabis sativa L.) produces an estimated 545 chemical compounds of different biogenetic classes. In addition to economic value, many of these phytochemicals have medicinal and physiological activity. The plant is most popularly known for its two most-prominent and most-studied secondary metabolites-Δ9-tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD). Both Δ9-THC and CBD have a wide therapeutic window across many ailments and form part of a class of secondary metabolites called cannabinoids-of which approximately over 104 exist. “Breast cancer is the leading cause of cancer-related death in women worldwide. In the last years, cannabinoids have gained attention in the clinical setting and clinical trials with cannabinoid-based preparations are underway. However, contradictory anti-tumour properties have also been reported. Thus, the elucidation of the molecular mechanisms behind their anti-tumour efficacy is crucial to better understand its therapeutic potential.
“Breast cancer is the leading cause of cancer-related death in women worldwide. In the last years, cannabinoids have gained attention in the clinical setting and clinical trials with cannabinoid-based preparations are underway. However, contradictory anti-tumour properties have also been reported. Thus, the elucidation of the molecular mechanisms behind their anti-tumour efficacy is crucial to better understand its therapeutic potential. “Cannabis was extensively utilized for its medicinal properties till the 19th century. A steep decline in its medicinal usage was observed later due to its emergence as an illegal recreational drug. Advances in technology and scientific findings led to the discovery of delta-9-tetrahydrocannabinol (THC), the primary psychoactive compound of cannabis, that further led to the discovery of endogenous cannabinoids system consisting of G-protein-coupled receptors – cannabinoid receptor 1 and cannabinoid receptor 2 along with their ligands, mainly anandamide and 2-arachidonoylglycerol. Endocannabinoid (EC) is shown to be a modulator not only for physiological functions but also for the immune system, endocrine network, and central nervous system. Medicinal research and meta-data analysis over the last few decades have shown a significant potential for both THC and cannabidiol (CBD) to exert palliative effects. People suffering from many forms of advanced stages of cancers undergo chemotherapy-induced nausea and vomiting followed by severe and chronic neuropathic pain and weight loss. THC and CBD exhibit effective analgesic, anxiolytic, and appetite-stimulating effect on patients suffering from cancer. Drugs currently available in the market to treat such chemotherapy-induced cancer-related ailments are Sativex (GW Pharmaceutical), Dronabinol (Unimed Pharmaceuticals), and Nabilone (Valeant Pharmaceuticals). Apart from exerting palliative effects, THC also shows promising role in the treatment of cancer growth, neurodegenerative diseases (multiple sclerosis and Alzheimer’s disease), and alcohol addiction and hence should be exploited for potential benefits. The current review discusses the nature and role of CB receptors, specific applications of cannabinoids, and major studies that have assessed the role of cannabinoids in cancer management.”
“Cannabis was extensively utilized for its medicinal properties till the 19th century. A steep decline in its medicinal usage was observed later due to its emergence as an illegal recreational drug. Advances in technology and scientific findings led to the discovery of delta-9-tetrahydrocannabinol (THC), the primary psychoactive compound of cannabis, that further led to the discovery of endogenous cannabinoids system consisting of G-protein-coupled receptors – cannabinoid receptor 1 and cannabinoid receptor 2 along with their ligands, mainly anandamide and 2-arachidonoylglycerol. Endocannabinoid (EC) is shown to be a modulator not only for physiological functions but also for the immune system, endocrine network, and central nervous system. Medicinal research and meta-data analysis over the last few decades have shown a significant potential for both THC and cannabidiol (CBD) to exert palliative effects. People suffering from many forms of advanced stages of cancers undergo chemotherapy-induced nausea and vomiting followed by severe and chronic neuropathic pain and weight loss. THC and CBD exhibit effective analgesic, anxiolytic, and appetite-stimulating effect on patients suffering from cancer. Drugs currently available in the market to treat such chemotherapy-induced cancer-related ailments are Sativex (GW Pharmaceutical), Dronabinol (Unimed Pharmaceuticals), and Nabilone (Valeant Pharmaceuticals). Apart from exerting palliative effects, THC also shows promising role in the treatment of cancer growth, neurodegenerative diseases (multiple sclerosis and Alzheimer’s disease), and alcohol addiction and hence should be exploited for potential benefits. The current review discusses the nature and role of CB receptors, specific applications of cannabinoids, and major studies that have assessed the role of cannabinoids in cancer management.” “The main aspects of severe COVID-19 disease pathogenesis include hyper-induction of proinflammatory cytokines, also known as ‘cytokine storm’, that precedes acute respiratory distress syndrome (ARDS) and often leads to death. COVID-19 patients often suffer from lung fibrosis, a serious and untreatable condition. There remains no effective treatment for these complications.
“The main aspects of severe COVID-19 disease pathogenesis include hyper-induction of proinflammatory cytokines, also known as ‘cytokine storm’, that precedes acute respiratory distress syndrome (ARDS) and often leads to death. COVID-19 patients often suffer from lung fibrosis, a serious and untreatable condition. There remains no effective treatment for these complications. “Inflammasomes are cytoplasmic inflammatory signaling protein complexes that detect microbial materials, sterile inflammatory insults, and certain host-derived elements. Inflammasomes, once activated, promote caspase-1-mediated maturation and secretion of pro-inflammatory cytokines, interleukin (IL)-1β and IL-18, leading to pyroptosis. Current advances in inflammasome research support their involvement in the development of chronic inflammatory disorders in contrast to their role in regulating innate immunity.
“Inflammasomes are cytoplasmic inflammatory signaling protein complexes that detect microbial materials, sterile inflammatory insults, and certain host-derived elements. Inflammasomes, once activated, promote caspase-1-mediated maturation and secretion of pro-inflammatory cytokines, interleukin (IL)-1β and IL-18, leading to pyroptosis. Current advances in inflammasome research support their involvement in the development of chronic inflammatory disorders in contrast to their role in regulating innate immunity. “Preclinical data suggest some cannabinoids may exert antitumour effects against glioblastoma (GBM). Safety and preliminary efficacy of nabiximols oromucosal cannabinoid spray plus dose-intense temozolomide (DIT) was evaluated in patients with first recurrence of GBM.
“Preclinical data suggest some cannabinoids may exert antitumour effects against glioblastoma (GBM). Safety and preliminary efficacy of nabiximols oromucosal cannabinoid spray plus dose-intense temozolomide (DIT) was evaluated in patients with first recurrence of GBM. “Glioblastomas (GBMs) are aggressive brain tumors with frequent genetic alterations in TP53 and PTEN tumor suppressor genes rendering resistance to standard chemotherapeutics. Cannabinoid type 1 and 2 (CB1/CB2) receptor expression in GBMs and antitumor activity of cannabinoids in glioma cells and animal models, raised promises for a targeted treatment of these tumors. The susceptibility of human glioma cells to CB2-agonists and their mechanism of action are not fully elucidated. We determined CB1 and CB2 expression in 14 low-grade and 21 high-grade tumor biopsies, GBM-derived primary cultures and established cell lines. The non-selective CB receptor agonist WIN55,212-2 (but not its inactive enantiomer) or the CB2-selective agonist JWH133 induced apoptosis in patient-derived glioma cultures and five established glioma cell lines despite p53 and/or PTEN deficiency. Growth inhibitory efficacy of cannabinoids correlated with CB1/CB2 expression (EC50 WIN55,212-2: 7.36-15.70 µM, JWH133: 12.15-143.20 µM). Treatment with WIN55,212-2 or JWH133 led to activation of the apoptotic mitochondrial pathway and DNA fragmentation. Synthetic cannabinoid action was associated with the induction of autophagy and knockdown of autophagy genes augmented cannabinoid-induced apoptotic cell death. The high susceptibility of human glioblastoma cells to synthetic cannabinoids, despite genetic defects contributing to apoptosis resistance, makes cannabinoids promising anti-glioma therapeutics.”
“Glioblastomas (GBMs) are aggressive brain tumors with frequent genetic alterations in TP53 and PTEN tumor suppressor genes rendering resistance to standard chemotherapeutics. Cannabinoid type 1 and 2 (CB1/CB2) receptor expression in GBMs and antitumor activity of cannabinoids in glioma cells and animal models, raised promises for a targeted treatment of these tumors. The susceptibility of human glioma cells to CB2-agonists and their mechanism of action are not fully elucidated. We determined CB1 and CB2 expression in 14 low-grade and 21 high-grade tumor biopsies, GBM-derived primary cultures and established cell lines. The non-selective CB receptor agonist WIN55,212-2 (but not its inactive enantiomer) or the CB2-selective agonist JWH133 induced apoptosis in patient-derived glioma cultures and five established glioma cell lines despite p53 and/or PTEN deficiency. Growth inhibitory efficacy of cannabinoids correlated with CB1/CB2 expression (EC50 WIN55,212-2: 7.36-15.70 µM, JWH133: 12.15-143.20 µM). Treatment with WIN55,212-2 or JWH133 led to activation of the apoptotic mitochondrial pathway and DNA fragmentation. Synthetic cannabinoid action was associated with the induction of autophagy and knockdown of autophagy genes augmented cannabinoid-induced apoptotic cell death. The high susceptibility of human glioblastoma cells to synthetic cannabinoids, despite genetic defects contributing to apoptosis resistance, makes cannabinoids promising anti-glioma therapeutics.”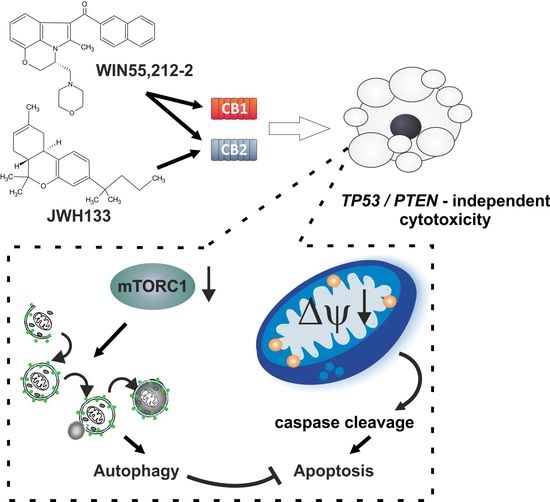
 “Glioblastoma is the most aggressive cancer among primary brain tumours. As with other cancers, the incidence of glioblastoma is increasing; despite modern therapies, the overall mean survival of patients post-diagnosis averages around 16 months, a figure that has not changed in many years. Cannabigerol (CBG) has only recently been reported to prevent the progression of certain carcinomas and has not yet been studied in glioblastoma. Here, we have compared the cytotoxic, apoptotic, and anti-invasive effects of the purified natural cannabinoid CBG together with CBD and THC on established differentiated glioblastoma tumour cells and glioblastoma stem cells. CBG and THC reduced the viability of both types of cells to a similar extent, whereas combining CBD with CBG was more efficient than with THC. CBD and CBG, both alone and in combination, induced caspase-dependent cell apoptosis, and there was no additive THC effect. Of note, CBG inhibited glioblastoma invasion in a similar manner to CBD and the chemotherapeutic temozolomide. We have demonstrated that THC has little added value in combined-cannabinoid glioblastoma treatment, suggesting that this psychotropic cannabinoid should be replaced with CBG in future clinical studies of glioblastoma therapy.”
“Glioblastoma is the most aggressive cancer among primary brain tumours. As with other cancers, the incidence of glioblastoma is increasing; despite modern therapies, the overall mean survival of patients post-diagnosis averages around 16 months, a figure that has not changed in many years. Cannabigerol (CBG) has only recently been reported to prevent the progression of certain carcinomas and has not yet been studied in glioblastoma. Here, we have compared the cytotoxic, apoptotic, and anti-invasive effects of the purified natural cannabinoid CBG together with CBD and THC on established differentiated glioblastoma tumour cells and glioblastoma stem cells. CBG and THC reduced the viability of both types of cells to a similar extent, whereas combining CBD with CBG was more efficient than with THC. CBD and CBG, both alone and in combination, induced caspase-dependent cell apoptosis, and there was no additive THC effect. Of note, CBG inhibited glioblastoma invasion in a similar manner to CBD and the chemotherapeutic temozolomide. We have demonstrated that THC has little added value in combined-cannabinoid glioblastoma treatment, suggesting that this psychotropic cannabinoid should be replaced with CBG in future clinical studies of glioblastoma therapy.”