“The transcription factor NFκB drives neoplastic progression of many cancers including primary brain tumors (glioblastoma; GBM). Precise therapeutic modulation of NFκB activity can suppress central oncogenic signalling pathways in GBM, but clinically applicable compounds to achieve this goal have remained elusive.
Methods: In a pharmacogenomics study with a panel of transgenic glioma cells we observed that NFκB can be converted into a tumor suppressor by the non-psychotropic cannabinoid Cannabidiol (CBD). Subsequently, we investigated the anti-tumor effects of CBD, which is used as an anticonvulsive drug (Epidiolex) in pediatric neurology, in a larger set of human primary GBM stem-like cells (hGSC). For this study we performed pharmacological assays, gene expression profiling, biochemical and cell-biological experiments. We validated our findings using orthotopic in vivo models and bioinformatics analysis of human GBM-datasets.
Results: We found that CBD promotes DNA binding of the NFκB subunit RELA and simultaneously prevents RELA-phosphorylation on serine-311, a key residue which permits genetic transactivation. Strikingly, sustained DNA binding by RELA lacking phospho-serine 311 was found to mediate hGSC cytotoxicity. Widespread sensitivity to CBD was observed in a cohort of hGSC defined by low levels of reactive oxygen-species (ROS), while high ROS-content in other tumors blocked CBD induced hGSC death. Consequently, ROS levels served as predictive biomarker for CBD-sensitive tumors.
Conclusions: This evidence demonstrates how a clinically approved drug can convert NFκB into a tumor suppressor and suggests a promising repurposing option for GBM-therapy.”
https://pubmed.ncbi.nlm.nih.gov/33864076/
https://academic.oup.com/neuro-oncology/advance-article/doi/10.1093/neuonc/noab095/6231710

 “The activity of a new, terpene-based formulation, code-named NT-VRL-1, against Human Coronavirus (HCoV) strain 229E was evaluated in human lung fibroblasts (MRC-5 cells), with and without the addition of cannabidiol (CBD). The main constituents in the terpene formulation used for the experiment were beta caryophyllene, eucalyptol, and citral. The tested formulation exhibited an antiviral effect when it was pre-incubated with the host cells prior to virus infection. The combination of NT-VRL-1 with CBD potentiated the antiviral effect better than the positive controls pyrazofurin and glycyrrhizin. There was a strong correlation between the quantitative results from a cell-viability assay and the cytopathic effect seen under the microscope after 72 h. To the best of our knowledge, this is the first report of activity of a combination of terpenes and CBD against a coronavirus.”
“The activity of a new, terpene-based formulation, code-named NT-VRL-1, against Human Coronavirus (HCoV) strain 229E was evaluated in human lung fibroblasts (MRC-5 cells), with and without the addition of cannabidiol (CBD). The main constituents in the terpene formulation used for the experiment were beta caryophyllene, eucalyptol, and citral. The tested formulation exhibited an antiviral effect when it was pre-incubated with the host cells prior to virus infection. The combination of NT-VRL-1 with CBD potentiated the antiviral effect better than the positive controls pyrazofurin and glycyrrhizin. There was a strong correlation between the quantitative results from a cell-viability assay and the cytopathic effect seen under the microscope after 72 h. To the best of our knowledge, this is the first report of activity of a combination of terpenes and CBD against a coronavirus.”

 “Breast cancer is the leading cause of cancer-related death in women worldwide. In the last years, cannabinoids have gained attention in the clinical setting and clinical trials with cannabinoid-based preparations are underway. However, contradictory anti-tumour properties have also been reported. Thus, the elucidation of the molecular mechanisms behind their anti-tumour efficacy is crucial to better understand its therapeutic potential.
“Breast cancer is the leading cause of cancer-related death in women worldwide. In the last years, cannabinoids have gained attention in the clinical setting and clinical trials with cannabinoid-based preparations are underway. However, contradictory anti-tumour properties have also been reported. Thus, the elucidation of the molecular mechanisms behind their anti-tumour efficacy is crucial to better understand its therapeutic potential. “The rapid spread of COVID-19 underscores the need for new treatments.
“The rapid spread of COVID-19 underscores the need for new treatments. “Inflammasomes are cytoplasmic inflammatory signaling protein complexes that detect microbial materials, sterile inflammatory insults, and certain host-derived elements. Inflammasomes, once activated, promote caspase-1-mediated maturation and secretion of pro-inflammatory cytokines, interleukin (IL)-1β and IL-18, leading to pyroptosis. Current advances in inflammasome research support their involvement in the development of chronic inflammatory disorders in contrast to their role in regulating innate immunity.
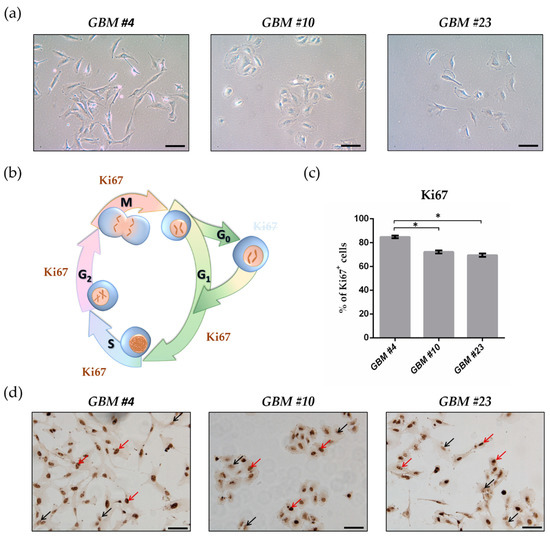
“Inflammasomes are cytoplasmic inflammatory signaling protein complexes that detect microbial materials, sterile inflammatory insults, and certain host-derived elements. Inflammasomes, once activated, promote caspase-1-mediated maturation and secretion of pro-inflammatory cytokines, interleukin (IL)-1β and IL-18, leading to pyroptosis. Current advances in inflammasome research support their involvement in the development of chronic inflammatory disorders in contrast to their role in regulating innate immunity. “Cannabidiol (CBD) has been shown to slow cancer cell growth and is toxic to human glioblastoma cell lines. Thus, CBD could be an effective therapeutic for glioblastoma.
“Cannabidiol (CBD) has been shown to slow cancer cell growth and is toxic to human glioblastoma cell lines. Thus, CBD could be an effective therapeutic for glioblastoma.
 “Endoplasmic reticulum (ER) stress is an imbalance between the ER’s protein-folding load and capacity. It can be induced by various physiological conditions, activating the unfolded protein response to re-establish homeostasis, promoting cell survival. Under severe or chronic stress, apoptosis is induced. Normal cells generally do not experience continuous ER stress induction. The stressful conditions experienced in the tumour microenvironment facilitates chronic ER stress and UPR activation, which plays a pivotal role in tumour survival.
“Endoplasmic reticulum (ER) stress is an imbalance between the ER’s protein-folding load and capacity. It can be induced by various physiological conditions, activating the unfolded protein response to re-establish homeostasis, promoting cell survival. Under severe or chronic stress, apoptosis is induced. Normal cells generally do not experience continuous ER stress induction. The stressful conditions experienced in the tumour microenvironment facilitates chronic ER stress and UPR activation, which plays a pivotal role in tumour survival.