
Recent data suggest that cannabinoids are involved in neuro-immuno-endocrine modulation of skin functioning, yet their effect on the features of dermatologic conditions is unclear. This article sought to review the mechanisms by which cannabinoids regulate skin functioning through the lens of relevance to treatment of dermatologic diseases looking at the effects of cannabinoids on a range of cellular activities and dermatologic conditions both in vitro and in vivo.
We identified studies demonstrating an inhibitory effect of cannabinoids on skin inflammation, proliferation, fibrosis, pain, and itch-biological mechanisms involved in the pathogenesis of many dermatologic conditions.
Cannabinoids have the potential to expand the therapeutic repertoire of a wide spectrum of skin disorders. Given their widespread unregulated use by the general public, basic and clinical studies are required to elucidate the effectiveness and long-term effects of topical and systemic cannabinoids in cutaneous disorders.”


 “The endocannabinoid system (ES) is a cell-signalling system widely distributed in biological tissues that includes endogenous ligands, receptors, and biosynthetic and hydrolysing machineries.
“The endocannabinoid system (ES) is a cell-signalling system widely distributed in biological tissues that includes endogenous ligands, receptors, and biosynthetic and hydrolysing machineries. “Sleep deprivation (SD) is a common feature in modern society. Prolonged sleep deprivation causes cognition deficits and depression-like behavior in the model of animal experiments.
“Sleep deprivation (SD) is a common feature in modern society. Prolonged sleep deprivation causes cognition deficits and depression-like behavior in the model of animal experiments. “Cannabinoids are a group of structurally heterogeneous but pharmacologically related compounds, including plant-derived cannabinoids, synthetic substances and endogenous cannabinoids, such as anandamide and 2-arachidonoylglycerol.
“Cannabinoids are a group of structurally heterogeneous but pharmacologically related compounds, including plant-derived cannabinoids, synthetic substances and endogenous cannabinoids, such as anandamide and 2-arachidonoylglycerol. “Cannabinoids are increasingly-used substances in the treatment of chronic pain, some neuropsychiatric disorders and more recently, skin disorders with an inflammatory component.
“Cannabinoids are increasingly-used substances in the treatment of chronic pain, some neuropsychiatric disorders and more recently, skin disorders with an inflammatory component. “Most of the drugs of abuse affect the brain by interacting with naturally expressed molecular receptors. Marihuana affects a series of receptors including
“Most of the drugs of abuse affect the brain by interacting with naturally expressed molecular receptors. Marihuana affects a series of receptors including  “Critically ill patients with sepsis require a multidisciplinary approach, as this situation implies multiorgan distress, with most of the bodily biochemical and cellular systems being affected by the condition. Moreover, sepsis is characterized by a multitude of biochemical interactions and by dynamic changes of the immune system. At the moment, there is a gap in our understanding of the cellular, genetic, and molecular mechanisms involved in sepsis.
“Critically ill patients with sepsis require a multidisciplinary approach, as this situation implies multiorgan distress, with most of the bodily biochemical and cellular systems being affected by the condition. Moreover, sepsis is characterized by a multitude of biochemical interactions and by dynamic changes of the immune system. At the moment, there is a gap in our understanding of the cellular, genetic, and molecular mechanisms involved in sepsis.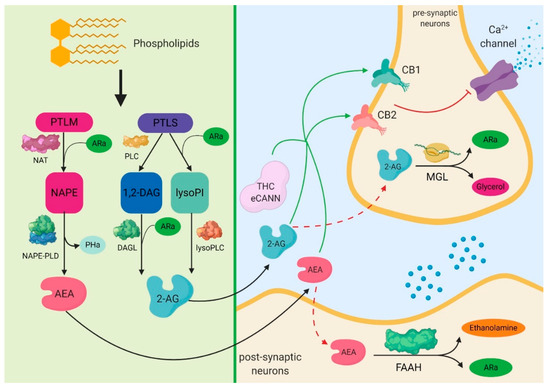
 “Human adipose tissue includes large quantities of mesenchymal stromal cells (atMSCs), which represent an abundant cell source for therapeutic applications in the field of regenerative medicine.
“Human adipose tissue includes large quantities of mesenchymal stromal cells (atMSCs), which represent an abundant cell source for therapeutic applications in the field of regenerative medicine.
 “Anticholinergic organophosphate (OP) agents act on the diverse serine hydrolases, thereby revealing unexpected biological effects. Epidemiological studies indicate a relationship between OP exposure and development of attention-deficit/hyperactivity disorder (ADHD)-like symptoms, whereas no plausible mechanism for the OP-induced ADHD has been established.
“Anticholinergic organophosphate (OP) agents act on the diverse serine hydrolases, thereby revealing unexpected biological effects. Epidemiological studies indicate a relationship between OP exposure and development of attention-deficit/hyperactivity disorder (ADHD)-like symptoms, whereas no plausible mechanism for the OP-induced ADHD has been established.