“Since antiquity, Cannabis has provoked enormous intrigue for its potential medicinal properties as well as for its unique pharmacological effects.
“Since antiquity, Cannabis has provoked enormous intrigue for its potential medicinal properties as well as for its unique pharmacological effects.
The elucidation of its major cannabinoid constituents, Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD), led to the synthesis of new cannabinoids (termed synthetic cannabinoids) to understand the mechanisms underlying the pharmacology of Cannabis.
These pharmacological tools were instrumental in the ultimate discovery of the endogenous cannabinoid system, which consists of CB1 and CB2 cannabinoid receptors and endogenously produced ligands (endocannabinoids), which bind and activate both cannabinoid receptors.
CB1 receptors mediate the cannabimimetic effects of THC and are highly expressed on presynaptic neurons in the nervous system, where they modulate neurotransmitter release. In contrast, CB2 receptors are primarily expressed on immune cells.
The endocannabinoids are tightly regulated by biosynthetic and hydrolytic enzymes. Accordingly, the endocannabinoid system plays a modulatory role in many physiological processes, thereby generating many promising therapeutic targets.
An unintended consequence of this research was the emergence of synthetic cannabinoids sold for human consumption to circumvent federal laws banning Cannabis use. Here, we describe research that led to the discovery of the endogenous cannabinoid system and show how knowledge of this system benefitted as well as unintentionally harmed human health.”

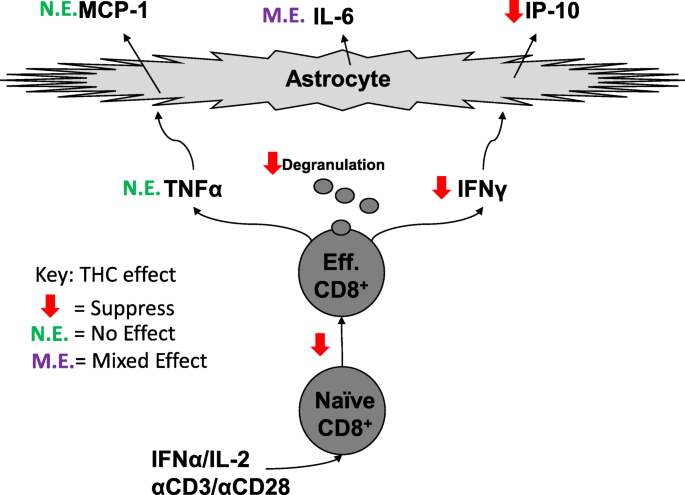 “CD8+ T cells can contribute to neuroinflammation by secretion of inflammatory cytokines like interferon γ (IFNγ) and tumor necrosis factor α (TNFα). Astrocytes, a glial cell in the brain, can be stimulated by IFNγ and TNFα to secrete the inflammatory cytokines, monocyte chemotactic protein 1 (MCP-1), interleukin 6 (IL-6), and interferon-γ inducible protein 10 (IP-10).
“CD8+ T cells can contribute to neuroinflammation by secretion of inflammatory cytokines like interferon γ (IFNγ) and tumor necrosis factor α (TNFα). Astrocytes, a glial cell in the brain, can be stimulated by IFNγ and TNFα to secrete the inflammatory cytokines, monocyte chemotactic protein 1 (MCP-1), interleukin 6 (IL-6), and interferon-γ inducible protein 10 (IP-10). “The present study investigated the effect of the lack of CB1 and CB2 receptors in mice ovarian morphology, folliculogenesis, oocyte retrieval, and oocyte maturation and evaluated the use of Δ9-tetrahydrocannabinol (THC) on oocyte in vitro maturation (IVM) by comparing classical IVM and two-step IVM by analyzing the meiotic competence of the oocytes and their evolution toward embryos.
“The present study investigated the effect of the lack of CB1 and CB2 receptors in mice ovarian morphology, folliculogenesis, oocyte retrieval, and oocyte maturation and evaluated the use of Δ9-tetrahydrocannabinol (THC) on oocyte in vitro maturation (IVM) by comparing classical IVM and two-step IVM by analyzing the meiotic competence of the oocytes and their evolution toward embryos. “Potential therapeutic actions of the cannabinoids delta-9-tetrahydrocannabinol (THC) and
“Potential therapeutic actions of the cannabinoids delta-9-tetrahydrocannabinol (THC) and  “To determine whether adjunctive dronabinol, a licensed form of delta-9-tetrahydrocannabinol, reduces opioid consumption when used off-label for managing acute pain following traumatic injury.
“To determine whether adjunctive dronabinol, a licensed form of delta-9-tetrahydrocannabinol, reduces opioid consumption when used off-label for managing acute pain following traumatic injury. “Excessive fear and anxiety, coupled with corticolimbic dysfunction, are core features of stress- and trauma-related psychopathology, such as posttraumatic stress disorder (PTSD).
“Excessive fear and anxiety, coupled with corticolimbic dysfunction, are core features of stress- and trauma-related psychopathology, such as posttraumatic stress disorder (PTSD).
 “Ratios of delta-9-tetrahydrocannabinol (THC) and
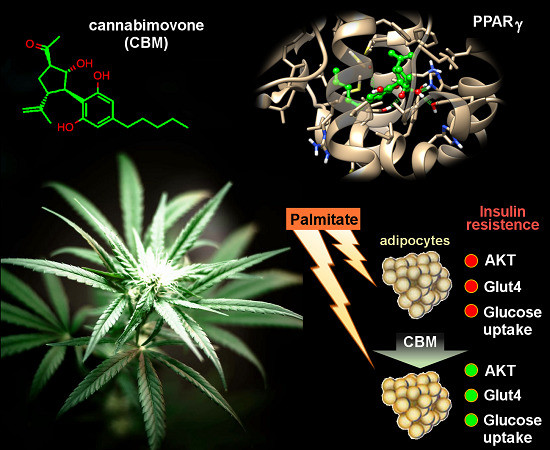
“Ratios of delta-9-tetrahydrocannabinol (THC) and  “Phytocannabinoids (pCBs) are a large family of meroterpenoids isolated from the plant Cannabis sativa. Δ9-Tetrahydrocannabinol (THC) and cannabidiol (CBD) are the best investigated phytocannabinoids due to their relative abundance and interesting bioactivity profiles. In addition to various targets, THC and CBD are also well-known agonists of peroxisome proliferator-activated receptor gamma (PPARγ), a nuclear receptor involved in energy homeostasis and lipid metabolism. In the search of new pCBs potentially acting as PPARγ agonists, we identified cannabimovone (CBM), a structurally unique abeo-menthane pCB, as a novel PPARγ modulator via a combined computational and experimental approach. The ability of CBM to act as dual PPARγ/α agonist was also evaluated. Computational studies suggested a different binding mode toward the two isoforms, with the compound able to recapitulate the pattern of H-bonds of a canonical agonist only in the case of PPARγ. Luciferase assays confirmed the computational results, showing a selective activation of PPARγ by CBM in the low micromolar range. CBM promoted the expression of PPARγ target genes regulating the adipocyte differentiation and prevented palmitate-induced insulin signaling impairment. Altogether, these results candidate CBM as a novel bioactive compound potentially useful for the treatment of insulin resistance-related disorders.”
“Phytocannabinoids (pCBs) are a large family of meroterpenoids isolated from the plant Cannabis sativa. Δ9-Tetrahydrocannabinol (THC) and cannabidiol (CBD) are the best investigated phytocannabinoids due to their relative abundance and interesting bioactivity profiles. In addition to various targets, THC and CBD are also well-known agonists of peroxisome proliferator-activated receptor gamma (PPARγ), a nuclear receptor involved in energy homeostasis and lipid metabolism. In the search of new pCBs potentially acting as PPARγ agonists, we identified cannabimovone (CBM), a structurally unique abeo-menthane pCB, as a novel PPARγ modulator via a combined computational and experimental approach. The ability of CBM to act as dual PPARγ/α agonist was also evaluated. Computational studies suggested a different binding mode toward the two isoforms, with the compound able to recapitulate the pattern of H-bonds of a canonical agonist only in the case of PPARγ. Luciferase assays confirmed the computational results, showing a selective activation of PPARγ by CBM in the low micromolar range. CBM promoted the expression of PPARγ target genes regulating the adipocyte differentiation and prevented palmitate-induced insulin signaling impairment. Altogether, these results candidate CBM as a novel bioactive compound potentially useful for the treatment of insulin resistance-related disorders.”