 “The endocannabinoid system (eCB) is a ubiquitous lipid signaling system composed of at least two receptors, their endogenous ligands, and the enzymes responsible for their synthesis and degradation. Within the brain, the eCB system is highly expressed in the hippocampus and controls basic biological processes, including neuronal proliferation, migration and differentiation, which are intimately linked with embryonal neurogenesis. Accumulated preclinical evidence has indicated that eCBs play a major role also in regulating adult neurogenesis. Increased cannabinoid receptor activity, either by increased eCB content or by pharmacological blockade of their degradation, produces neurogenic effects alongside rescue of phenotypes in animal models of different psychiatric and neurological disorders. Therefore, in the light of the higher therapeutic potential of adult neurogenesis compared to the embryonic one, here we sought to summarize the most recent evidence pointing towards a neurogenic role for eCBs in the adult brain, both under normal and pathological conditions.”
“The endocannabinoid system (eCB) is a ubiquitous lipid signaling system composed of at least two receptors, their endogenous ligands, and the enzymes responsible for their synthesis and degradation. Within the brain, the eCB system is highly expressed in the hippocampus and controls basic biological processes, including neuronal proliferation, migration and differentiation, which are intimately linked with embryonal neurogenesis. Accumulated preclinical evidence has indicated that eCBs play a major role also in regulating adult neurogenesis. Increased cannabinoid receptor activity, either by increased eCB content or by pharmacological blockade of their degradation, produces neurogenic effects alongside rescue of phenotypes in animal models of different psychiatric and neurological disorders. Therefore, in the light of the higher therapeutic potential of adult neurogenesis compared to the embryonic one, here we sought to summarize the most recent evidence pointing towards a neurogenic role for eCBs in the adult brain, both under normal and pathological conditions.”
https://www.ncbi.nlm.nih.gov/pubmed/31864101
“The endocannabinoid system is involved in all aspects of the biology of neural stem cells. Selective CB1 and CB2 agonism produces pro-neurogenic effects in different models of brain insults. Further research is needed to characterize the eCB system as a new druggable target for neurogenesis-related diseases.”
https://www.sciencedirect.com/science/article/pii/S1471489219301122?via%3Dihub


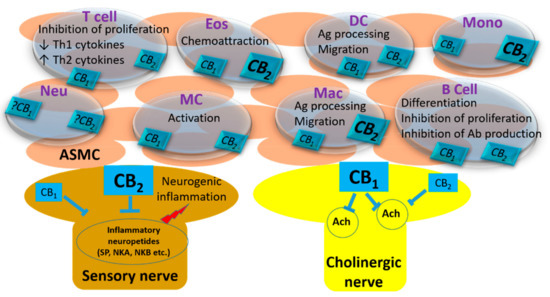
 “Even a brief exposure to severe stress strengthens synaptic connectivity days later in the amygdala, a brain area implicated in the affective symptoms of stress-related psychiatric disorders. However, little is known about the synaptic signaling mechanisms during stress that eventually culminate in its delayed impact on the amygdala. Hence, we investigated early stress-induced changes in amygdalar synaptic signaling in order to prevent its delayed effects.
“Even a brief exposure to severe stress strengthens synaptic connectivity days later in the amygdala, a brain area implicated in the affective symptoms of stress-related psychiatric disorders. However, little is known about the synaptic signaling mechanisms during stress that eventually culminate in its delayed impact on the amygdala. Hence, we investigated early stress-induced changes in amygdalar synaptic signaling in order to prevent its delayed effects. “This review focuses on the possible roles of phytocannabinoids, synthetic cannabinoids, endocannabinoids, and “transient receptor potential cation channel, subfamily V, member 1” (TRPV1) channel blockers in epilepsy treatment.
“This review focuses on the possible roles of phytocannabinoids, synthetic cannabinoids, endocannabinoids, and “transient receptor potential cation channel, subfamily V, member 1” (TRPV1) channel blockers in epilepsy treatment. “The increasing medicinal use of
“The increasing medicinal use of  “In drug development, the creation of highly selective ligands has been unsuccessful for the treatment of central nervous system disorders.
“In drug development, the creation of highly selective ligands has been unsuccessful for the treatment of central nervous system disorders.
 “Irritable bowel syndrome (IBS) is one of the most common functional gastrointestinal (GI) disorders characterized by pain and impaired bowel movements. Currently available drugs show limited efficacy.
“Irritable bowel syndrome (IBS) is one of the most common functional gastrointestinal (GI) disorders characterized by pain and impaired bowel movements. Currently available drugs show limited efficacy. “Evidence suggests that activation of the endocannabinoid system offers cardioprotection.
“Evidence suggests that activation of the endocannabinoid system offers cardioprotection.